Filed pursuant to Rule 253(g)(2)
File No. 024-11602
Supplement No. 1 DATED DECEMBER 14, 2021
(to the Offering Circular dated December 13, 2021)
RespireRx Pharmaceuticals Inc.
126 Valley Road, Suite C
Glen Rock, New Jersey 07452
(201) 444-4947
This Offering Circular Supplement No. 1 (“Supplement No. 1”) supplements and amends the offering circular of RespireRx Pharmaceuticals, Inc. (the “Company”) dated December 13, 2021 the “Offering Circular”), relating to Company’s Tier 2 offering under Regulation A of Section 3(6) of the Securities Act of 1933, as amended, of up to 375,000,000 shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at an offering price of $0.02 per share, for a maximum offering amount of $7,500,000 (the “Offering”). This Supplement No. 1 should be read in conjunction with the Offering Circular and is qualified by reference to the Offering Circular except to the extent that the information in this Supplement No. 1 supersedes the information contained in the Offering Circular.
Since December 13, 2021, the Company has submitted a form 8-K with three exhibits related to a press release and two slide decks to be presented at upcoming investor conferences.
This Supplement No.1 attaches the Current Report on Form 8-K that the Company filed with the U.S. Securities and Exchange Commission on December 14, 2021.
Investing in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading “Risk Factors” beginning on page 10 of the Offering Circular, and under similar headings in any amendments or supplements to the Offering Circular.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of the Offering Circular or this Supplement No. 1. Any representation to the contrary is a criminal offense.
The date of this Supplement No. 1 is December 14, 2021
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
Current Report
Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): December 10, 2021
RESPIRERX PHARMACEUTICALS INC.
(Exact name of registrant as specified in its charter)
| Delaware | 1-16467 | 33-0303583 | ||
(State or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S Employer Identification No.) |
126 Valley Road, Suite C Glen Rock, New Jersey |
07452 | |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including area code: (201) 444-4947
(Former name or former address, if changed since last report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| N/A | N/A | N/A |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01. Regulation FD Disclosure.
RespireRx Pharmaceuticals Inc. (the “Company”) announced today, that its Chief Financial Officer, Jeff Margolis, will be presenting at the Life Sciences Virtual Investor Conference (“LSVIC”) on December 16, 2021 at 10:00 am EST. A copy of the press release describing the conference and the Company’s participation is attached as Exhibit 99.1. In addition, the presentation materials in the form of the slide deck to be presented is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
In the same press release that is Exhibit 99.1, RespireRx announced that the Company’s video presentation for the Biotech Showcase™ was made available by the Biotech Showcase™ on Friday, December 10, 2021 to registered participants. The slide deck, both in video and pdf format (without video), will be available on the Company’s website at www.respirerx.com and the pdf format (without video) is attached as Exhibit 99.3 to this Current Report on Form 8-K and is incorporated herein by reference.
The press release and the slide decks that are Exhibits 99.1, 99.2 and 99.3 include certain forward-looking information.
The information in this Item 7.01 and the documents attached as Exhibit 99.1, 99.2 and 99.3 are being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities and Exchange Act of 1934, as amended (the “Exchange Act”), nor otherwise subject to the liabilities of that section, nor incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such a filing.
Item 9.01 Financial Statements and Exhibits
(d) Exhibits.
| Exhibit Number | Exhibit Description | |
| 99.1 | RespireRx Pharmaceuticals Inc. Press Release | |
99.2 |
RespireRx Pharmaceuticals Inc. Slide deck for Life Sciences Virtual Investor Conference | |
| 99.3 | RespireRx Pharmaceuticals Inc. Slide deck for Biotech Showcase™ | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Date: December 14, 2021 | RESPIRERX PHARMACEUTICALS INC. | |
| (Registrant) | ||
| By: | /s/ Jeff E. Margolis | |
Jeff E. Margolis | ||
| SVP, CFO, Secretary and Treasurer | ||

RespireRx Pharmaceuticals Inc. to Present at Upcoming Virtual Life Sciences Investor Conference and Biotech Showcase™
Glen Rock, N.J., December 14, 2021 /Globe Newswire – RespireRx Pharmaceuticals Inc. (OTCQB: RSPI) (“RespireRx” or the “Company”), with a mission to discover and develop innovative and revolutionary treatments to combat diseases caused by disruption of neuronal signaling, is pleased to announce that its Chief Financial Officer, Jeff Margolis, will be presenting at the Life Sciences Virtual Investor Conference (“LSVIC”) on December 16, 2021 at 10:00 am EST, and its President and CEO, Tim Jones will be presenting via pre-recorded video at the virtual Biotech Showcase™ taking place January 17 – 19, 2022.
Life Sciences Virtual Investor Conference
At the LSVIC, Mr. Margolis will be sharing the Company’s vision, its platform and program areas and answering audience questions. The slide deck used during the presentation will be available on the Company’s website and as furnished (not filed) to the SEC as an exhibit to our Current Report on Form 8-K available at www.sec.gov
Virtual Investor Conferences® is a proprietary investor conference series and is part of the OTC Market Group’s suite of investor relations services. These conferences are quarterly events for public and private companies, investors, and industry professionals from around the world. The LSVIC is a day-long virtual event with live company presentations and interactive discussions.
Individual investors, institutional investors, advisors, and analysts are invited to attend. The program opens at 9:15 am EST on Thursday, December 16th with the first live webcast at 9:30 am EST. Mr. Margolis is presenting at 10:00 am.
Interested parties may register at https://bit.ly/3Go96iH.
It is recommended that investors pre-register and run the online system check to expedite participation and receive event updates. There is no cost to log-in, attend the live presentations or ask questions.
Biotech Showcase™
The Company’s video presentation was made available by the Biotech Showcase™ on Friday, December 10, 2021 and may be viewed at:
https://partneringone.informaconnect.com/event/716/company/615/content/13
by registered participants. The slide deck, both in video and pdf format (without video), will be available on the Company’s website at www.respirerx.com and as a furnished (not filed) exhibit to our Current Report on Form 8-K available at www.sec.gov. Because the Company is participating in the virtual portion of the Biotech Showcase™ and the presentation has been pre-recorded, there is no set schedule for presenting companies.
The in-person portion of the Biotech Showcase™ is taking place from January 10 – 12, 2022 in San Francisco, CA and virtual portion is taking place from January 17 - 19, 2022. Mr. Jones discusses the basis of the science behind RespireRx’s portfolio of clinical candidates, the Company’s short-term objectives and the potential global commercial opportunities that could be realized for the Company’s novel and differentiated suite of products, which address a number of neurological and psychiatric disorders for which there are currently quite limited and inferior treatment options.
The video presentation will also be played as follows during the in-person event and will be viewable by Biotech Showcase™ registered participants:
Date: Tuesday, January 11, 2022
Time: 11:30 am PST (2:30 pm EST)
Track: Yosemite A (Ballroom Level)
Biotech Showcase™ and its family of events, China Showcase, Digital Medicine & Medtech Showcase and Seed Showcase, are investor conferences devoted to providing private and micro-mid-cap biotechnology companies an opportunity to present to and meet with investors and biopharmaceutical executives. Featuring over 400 curated presenting companies from around the world, from seed-stage companies to public, multi-national companies, from platform technology to therapeutic areas to digital health and device and diagnostics, the event attracts investors representing over USD 400 Billion in capital, and highly motivated strategic partners in the life science ecosystem.
Biotech Showcase™ is being delivered in a new hybrid format. In-person components will take place January 10–12, 2022 at the Hilton San Francisco Union Square in San Francisco, CA. Virtual event components will follow the week after, January 17–19, 2022, hosted on partneringONE® for a seamless digital experience.
About RespireRx Pharmaceuticals Inc.
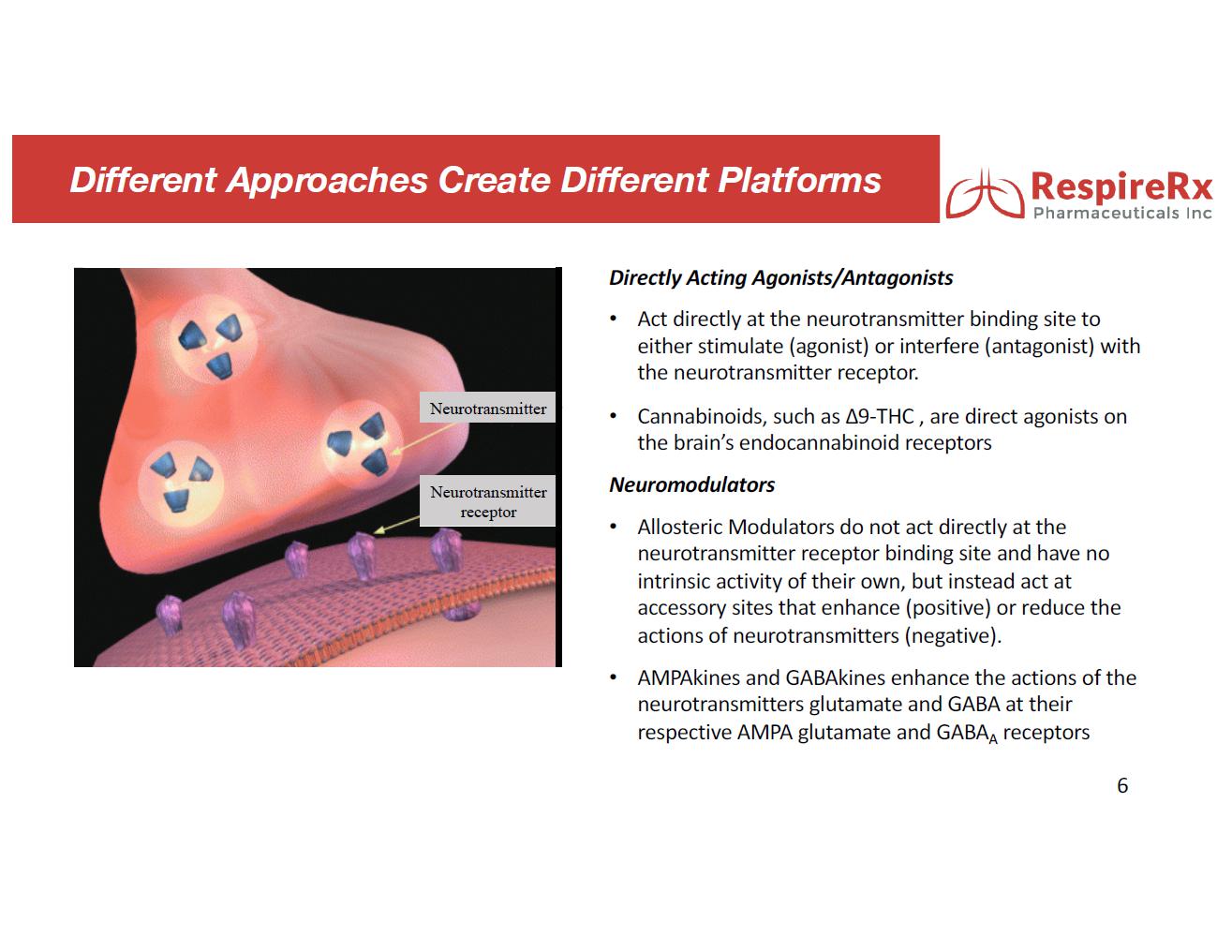
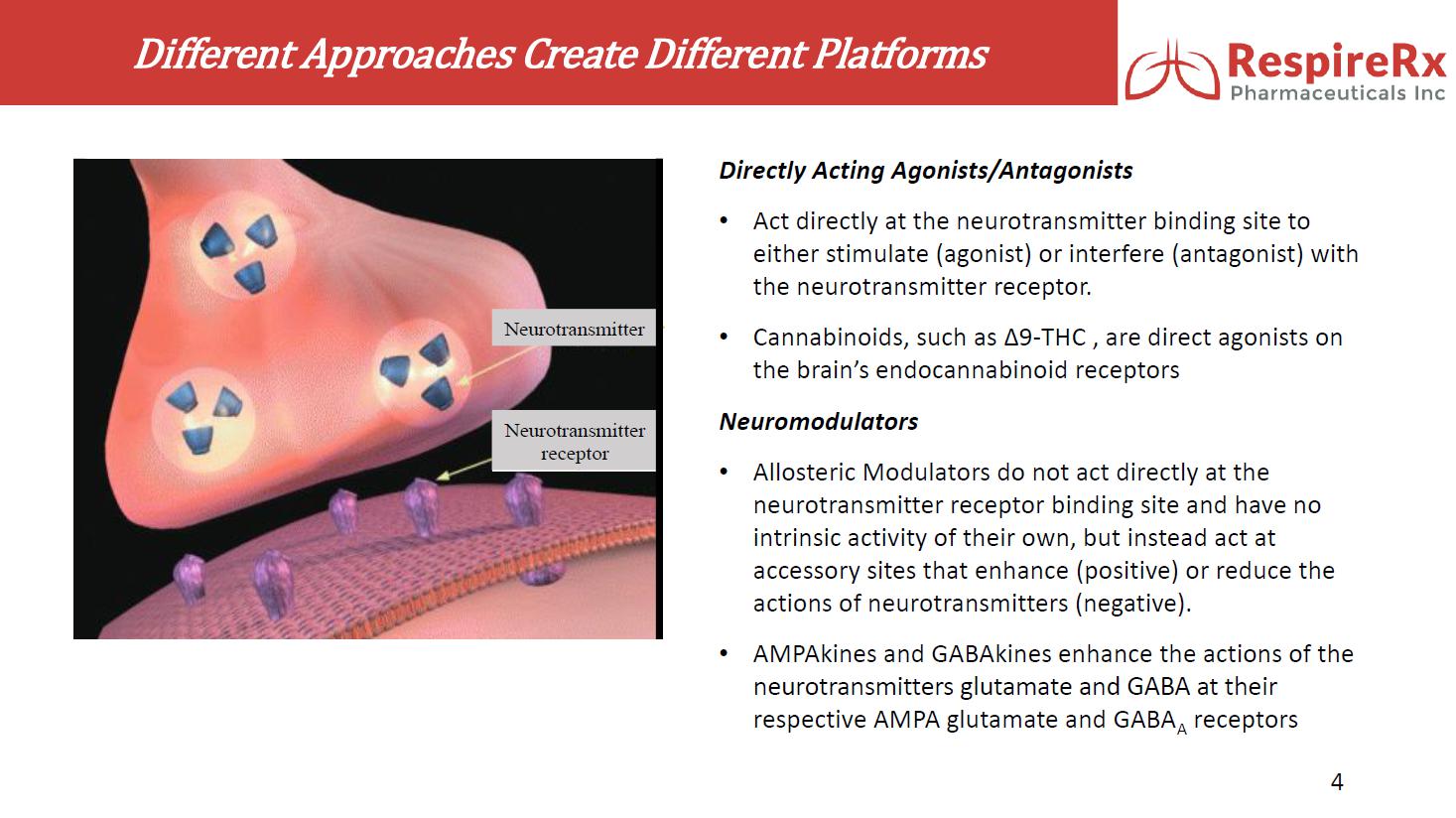
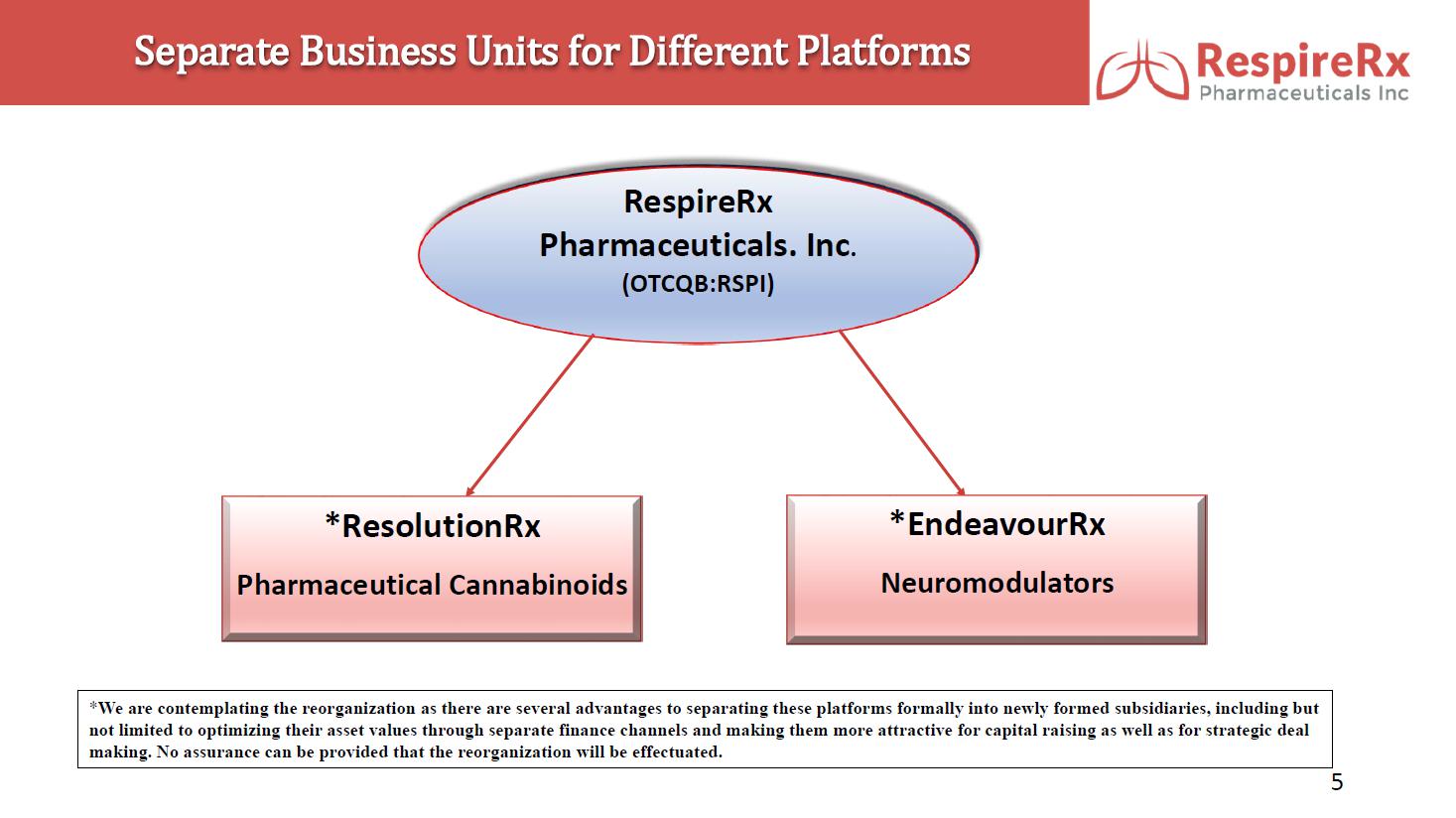
RespireRx Pharmaceuticals Inc. is a leader in the discovery and development of medicines for the treatment of psychiatric and neurological disorders, with a focus on treatment options that address conditions affecting millions of people, but for which there are few or poor treatment options, including obstructive sleep apnea (“OSA”), attention deficit hyperactivity disorder (“ADHD”), epilepsy, chronic pain and recovery from spinal cord injury (“SCI”), as well as certain neurological orphan diseases. RespireRx is developing a pipeline of new and re-purposed drug products based on our broad patent portfolios for two drug platforms: (i) pharmaceutical cannabinoids, which include dronabinol, a synthetic form of ∆9-tetrahydrocannabinol (“Δ9-THC”) that acts upon the nervous system’s endogenous cannabinoid receptors and (ii) neuromodulators, which include AMPAkines and GABAkines, proprietary chemical entities that positively modulate (positive allosteric modulators or “PAMs”) AMPA-type glutamate receptors and GABAA receptors, respectively.
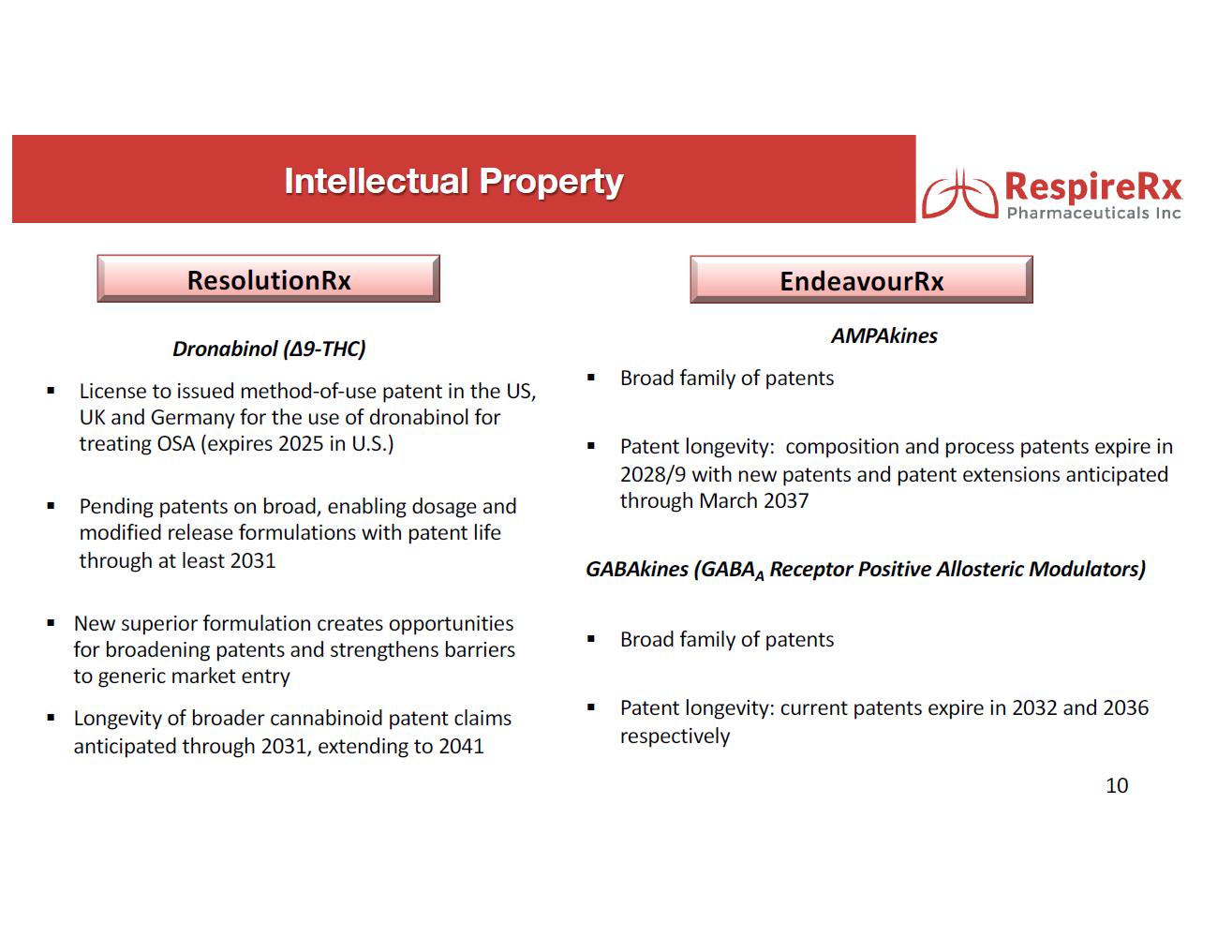
The Company holds exclusive licenses and owns patents and patent applications or rights thereto for certain families of chemical compounds that claim the chemical structures and their uses in the treatment of a variety of disorders, as well as claims for novel uses of known drugs.
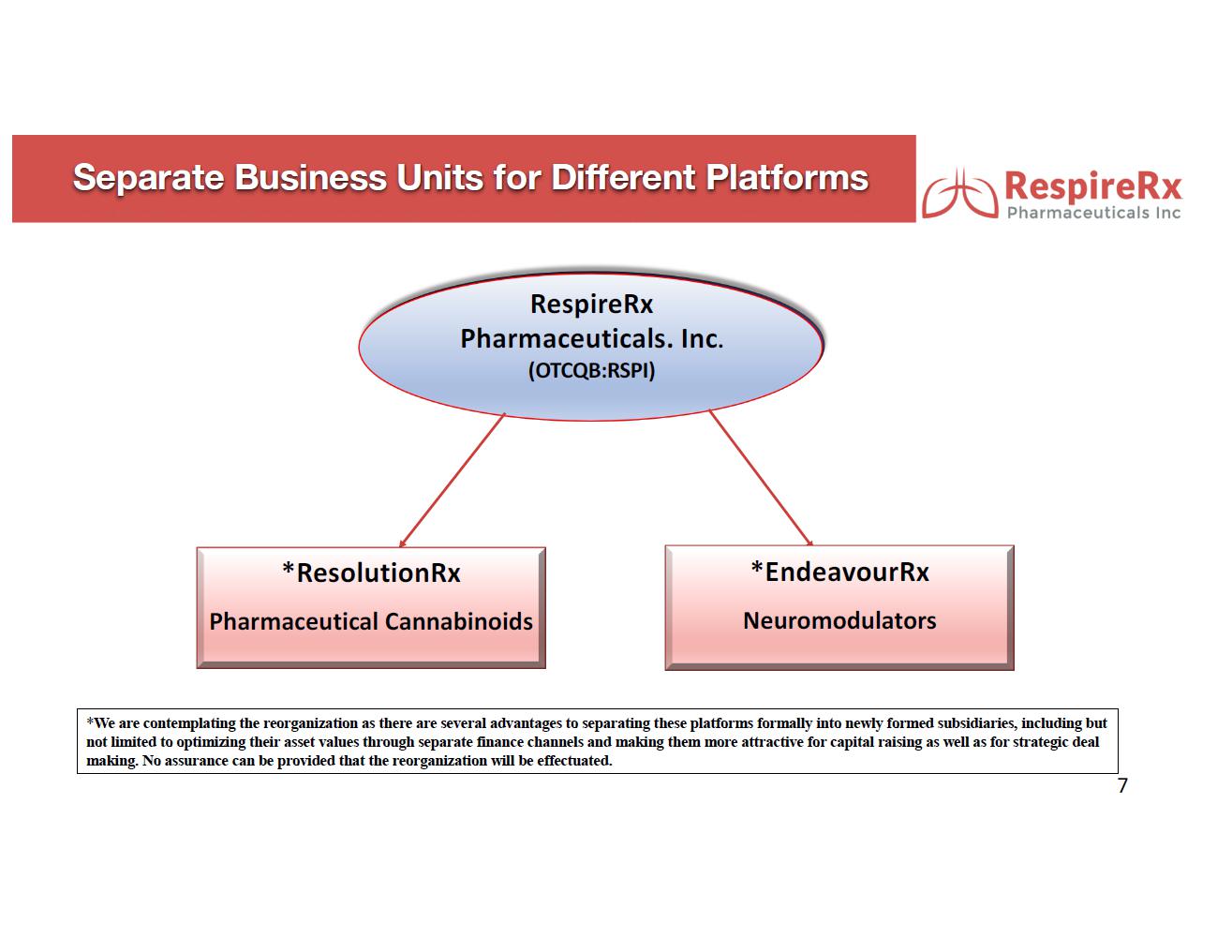
ResolutionRx: Pharmaceutical Cannabinoids.
Dronabinol. RespireRx is developing dronabinol, ∆-9-THC, a synthetic version of the naturally occurring substance in the cannabis plant, for the treatment of OSA, a serious respiratory disorder that impacts an estimated 29.4 million people in the United States according to the American Academy of Sleep Medicine (“AASM”), published in August 2016. OSA has been linked to increased risk for hypertension, heart failure, depression, and diabetes, and has an annual economic cost in the United States of $162 billion according to the AASM. There are no approved drug treatments for OSA.
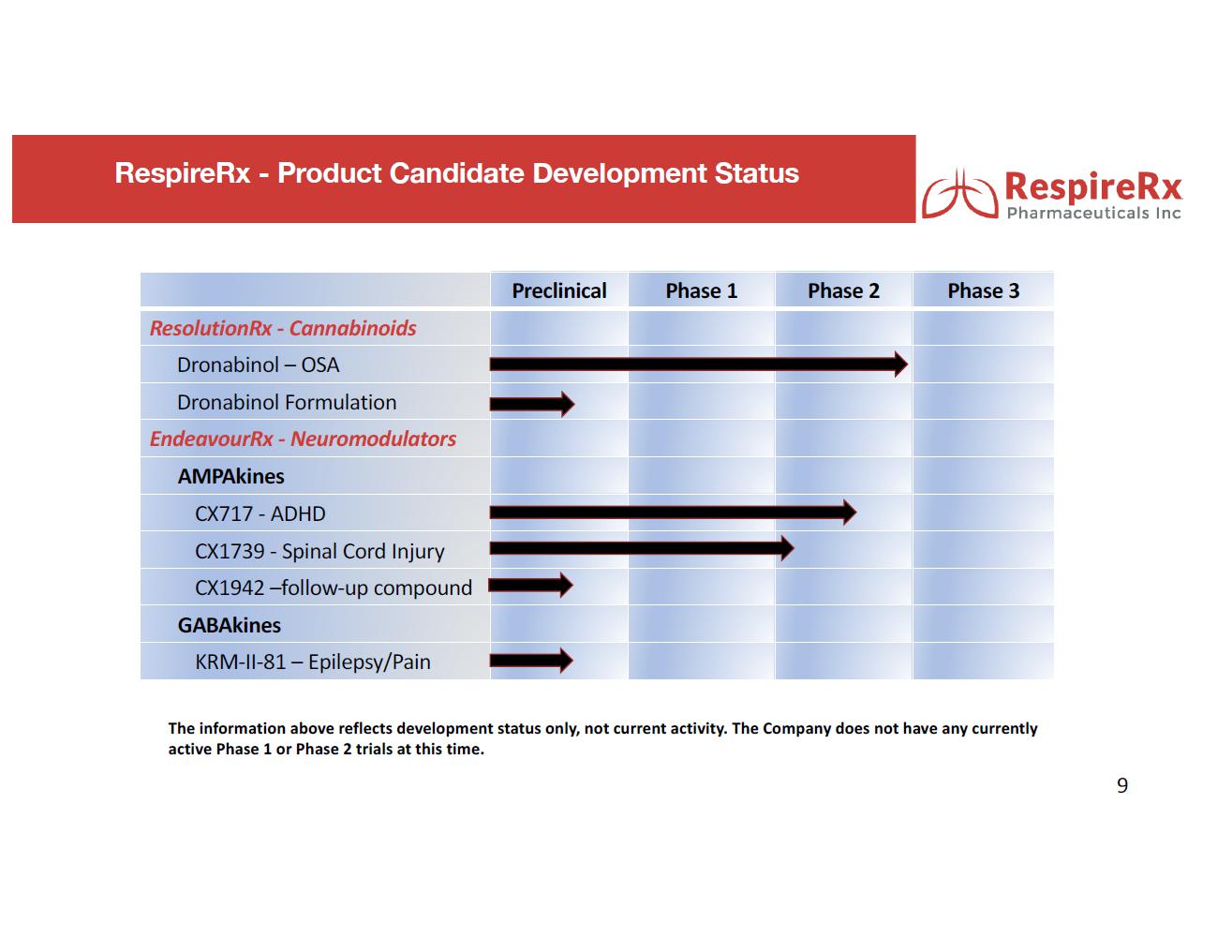
Two Phase 2 clinical trials have been completed demonstrating the ability of dronabinol to significantly reduce the symptoms of OSA and, subject to raising sufficient financing (of which no assurance can be provided) and pending the outcome of an intended meeting with the FDA, RespireRx believes that it will be able to commence a pharmacokinetic study for a recently discovered and to-be-developed formulation followed by a Phase 3 clinical study for the treatment of OSA with the new formulation. Because dronabinol is already FDA approved for the treatment of AIDS related anorexia and chemotherapy induced nausea and vomiting, the Company further believes that its re-purposing strategy would only require approval by the FDA of a 505(b)(2) new drug application (“NDA”), an efficient regulatory pathway that allows the use of publicly available data.
EndeavourRx: Neuromodulators
GABAkines. Under a License Agreement with the University of Wisconsin-Milwaukee Research Foundation, Inc. (“UWMRF”), RespireRx has licensed rights to certain selectively acting GABAkines because of their ability to selectively amplify inhibitory neurotransmission at a highly specific, subset of GABAA receptors, thus producing a unique efficacy profile with reduced side effects. Preclinical studies have documented their efficacy in a broad array of animal models of interrelated neurological and psychiatric disorders including epilepsy, pain, anxiety, and depression in the absence of or with greatly reduced propensity to produce sedation, motor-impairment, tolerance, dependence and abuse. The Company currently is focusing on developing KRM-II-81 for the treatment of epilepsy and pain.
KRM-II-81 has displayed a high degree of anti-convulsant activity in a broad range of preclinical studies, including in treatment resistant and pharmaco-resistant models. Not only was KRM-II-81 highly effective in these models, but pharmaco-resistance or tolerance did not develop to its anti-convulsant properties. These latter results are particularly important because pharmaco-resistance occurs when medications that once controlled seizures lose efficacy as a result of chronic use and it is a principle reason some epileptic patients require brain surgery to control their seizures. In support of its potential clinical efficacy, translational studies have demonstrated the ability of KRM-II-81 to dramatically reduce epileptiform electrical activity when administered in situ to brain slices excised from treatment resistant epileptic patients undergoing surgery.
In addition, KRM-II-81 has displayed a high degree of analgesic activity in a broad range of preclinical studies. In intact animal models of acute and chronic pain, the analgesic efficacy of KRM-II-81 was comparable to or greater than commonly used analgesics. At the same time, KRM-II-81 did not display side effects such as sedation and motor impairment, but even more importantly, it did not produce tolerance, dependence, respiratory depression or behavioral changes indicative of abuse liability, which are produced by opioid narcotics and are at the heart of the opioid epidemic.
AMPAkines. Through an extensive translational research effort from the cellular level through Phase 2 clinical trials, the Company has developed a family of novel, low impact AMPAkines, including CX717, CX1739 and CX1942 that may have clinical application in the treatment of CNS-driven neurobehavioral and cognitive disorders, spinal cord injury, neurological diseases, and certain orphan indications. Our lead clinical compounds, CX717 and CX1739, have successfully completed multiple Phase 1 safety trials. Both compounds have also completed Phase 2 proof of concept trials demonstrating target engagement, by antagonizing the ability of opioids to induce respiratory depression.
AMPAkines have demonstrated positive activity in animal models of ADHD, results that have been extended translationally into statistically significant improvement of symptoms observed in a Phase 2 human clinical trial of CX717 in adults with ADHD. Statistically significant therapeutic effects were observed within one week. We believe AMPAkines may represent a novel, non-stimulant treatment for ADHD with a more rapid onset of action than alternative non stimulants, such as Straterra® (atomoxetine), and without the drawbacks of amphetamine-type stimulants.
In a series of important studies funded by grants from the National Institutes of Health and published in a number of peer reviewed articles, Dr. David Fuller (University of Florida), a long-time RespireRx collaborator, has demonstrated the ability of CX1739 and CX717, the Company’s lead AMPAkines, to improve motor nerve activity and muscle function in animal models of spinal cord injury (SCI).
Additional information about RespireRx and the matters discussed herein can be obtained on the Company’s web-site at www.RespireRx.com or in the Company’s filings with the Securities and Exchange Commission at www.sec.gov.
Not a Securities Offering or Solicitation
This communication shall not constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sales of securities in any jurisdiction in which such offer, solicitation or sale of securities would be unlawful before registration or qualification under the laws of such jurisdiction.
Cautionary Note Regarding Forward-Looking Statements
This press release contains certain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”) and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and the Company intends that such forward-looking statements be subject to the safe harbor created thereby. These might include statements regarding the Company’s future plans, targets, estimates, assumptions, financial position, business strategy and other plans and objectives for future operations, and assumptions and predictions about research and development efforts, including, but not limited to, preclinical and clinical research design, execution, timing, costs and results, future product demand, supply, manufacturing, costs, marketing and pricing factors.
In some cases, forward-looking statements may be identified by words including “assumes,” “could,” “ongoing,” “potential,” “predicts,” “projects,” “should,” “will,” “would,” “anticipates,” “believes,” “intends,” “estimates,” “expects,” “plans,” “contemplates,” “targets,” “continues,” “budgets,” “may,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words, and such statements may include, but are not limited to, statements regarding (i) future research plans, expenditures and results, (ii) potential collaborative arrangements, (iii) the potential utility of the Company’s products candidates, (iv) reorganization plans, and (v) the need for, and availability of, additional financing. Forward-looking statements are based on information available at the time the statements are made and involve known and unknown risks, uncertainties and other factors that may cause our results, levels of activity, performance or achievements to be materially different from the information expressed or implied by the forward-looking statements in this press release.
These factors include but are not limited to, regulatory policies or changes thereto, available cash, research and development results, issuance of patents, competition from other similar businesses, interest of third parties in collaborations with us, and market and general economic factors, and other risk factors disclosed in “Item 1A. Risk Factors” in the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2020, as filed with the SEC on April 15, 2021 (the “2020 Form 10-K”).
You should read these risk factors and the other cautionary statements made in the Company’s filings as being applicable to all related forward-looking statements wherever they appear in this press release. We cannot assure you that the forward-looking statements in this press release will prove to be accurate and therefore prospective investors, as well as potential collaborators and other potential stakeholders, are encouraged not to place undue reliance on forward-looking statements. You should read this press release completely. Other than as required by law, we undertake no obligation to update or revise these forward-looking statements, even though our situation may change in the future.
We caution investors, as well as potential collaborators and other potential stakeholders, not to place undue reliance on any forward-looking statement that speaks only as of the date made and to recognize that forward-looking statements are predictions of future results, which may not occur as anticipated. Actual results could differ materially from those anticipated in the forward-looking statements and from historical results, due to the risks and uncertainties described in the 2020 Form 10-K and in this press release, as well as others that we may consider immaterial or do not anticipate at this time. These forward-looking statements are based on assumptions regarding the Company’s business and technology, which involve judgments with respect to, among other things, future scientific, economic, regulatory and competitive conditions, collaborations with third parties, and future business decisions, all of which are difficult or impossible to predict accurately and many of which are beyond the Company’s control. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. Our expectations reflected in our forward-looking statements can be affected by inaccurate assumptions that we might make or by known or unknown risks and uncertainties, including those described in the 2020 Form 10-K and in this press release. These risks and uncertainties are not exclusive and further information concerning us and our business, including factors that potentially could materially affect our financial results or condition, may emerge from time to time.
For more information about the risks and uncertainties the Company faces, see “Item 1A. Risk Factors” in our 2020 Form 10-K. Forward-looking statements speak only as of the date they are made. The Company does not undertake and specifically declines any obligation to update any forward-looking statements or to publicly announce the results of any revisions to any statements to reflect new information or future events or developments. We advise investors, as well as potential collaborators and other potential stakeholders, to consult any further disclosures we may make on related subjects in our annual reports on Form 10-K and other reports that we file with or furnish to the SEC.
Company Contact:
Jeff Margolis
Senior Vice President, Chief Financial Officer, Treasurer and Secretary
Telephone: (917) 834-7206
E-mail: jmargolis@respirerx.com
RespireRx Pharmaceuticals Inc.
126 Valley Road,
Suite C,
Glen Rock, NJ 07452
www.respirerx.com